electroCore Receives 510(k) Clearance for gammaCore-S® (non-invasive vagus nerve stimulator) for the Acute Treatment of Pain Associated with Episodic Cluster Headache in Adult Patients - electroCore

electroCore Receives 510(k) Clearance for gammaCore-S® (non-invasive vagus nerve stimulator) for the Acute Treatment of Pain Associated with Episodic Cluster Headache in Adult Patients - electroCore
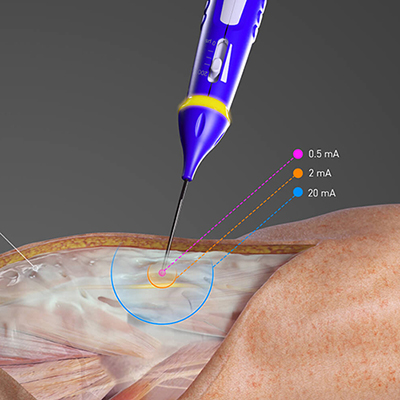
Regulatory clearance marks most recent achievement in new device being available to the approximately 400,000 Americans living with episodic cluster headaches Basking Ridge, NJ, June 15, 2017 – electroCore, a neuroscience and technology company dedicated to improving patient outcomes through technological advancement, announced today that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance to market a newer version of gammaCore® (non-invasive vagus nerve stimulator) – the gammaCore-S® – for the acute treatment of pain associated with episodic cluster headache (eCH) in adult patients. gammaCore transmits a mild electrical stimulation to the vagus nerve through the skin to block the pain signals that cause eCH. “This clearance represents a significant milestone for electroCore as we are now one step closer to bringing gammaCore to patients in the U.S.,” said Francis R. Amato, Chief Executive Officer of electroCore. “By being able to market this version of gammaCore in the U.S., adult patients will now have access to a more user-friendly device, with the latest technology to treat pain associated with episodic cluster headache.” gammaCore was released by the FDA for the acute treatment of pain associated with eCH in adult patients on April 14, 2017. gammaCore-S has

electroCore's non-invasive vagus nerve stimulation device shown to

ElectroCore launches gammaCore Sapphire migraine treatment device

Review of Non-Invasive Vagus Nerve Stimulation (gammaCore

Review of Non-Invasive Vagus Nerve Stimulation (gammaCore
Try gammaCore

In The News electroCore

gammaCore®, the First Non-Invasive Vagus Nerve Stimulator Applied

gammaCore Personal Non-Invasive VNS Cleared for Episodic Cluster

FDA Releases gammaCore®, the First Non-Invasive Vagus Nerve